Magnesium batteries safer than lithium?
on
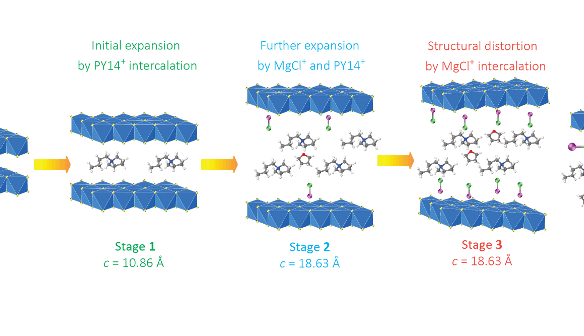
The research team has published their findings in the Nature Communications journal. Dr Yao and his team have succeeded in developing a new cathode that dramatically increases the battery’s energy density. According to post doctoral fellow Dr Hyun Yoo, co-author of the paper, it had previously been assumed that the magnesium-chloride bond needed to be broken before insertion of magnesium into the host material. The new battery stores energy by direct insertion of magnesium monochloride ions (MgCl) into the host material, For example, titanium disulfide (TiS2). As a result, the diffusion is stronger than in previous battery designs.
The researchers also achieved a capacitance density of 400 mAh/g - a fourfold increase compared to previous magnesium battery designs. By comparison lithium-ion batteries reach only about 200 mAh/g. The cell voltage of a magnesium cell is however only 1 V which is significantly less than lithium. Magnesium is a more abundant material and therefore less expensive than Lithium and is not prone to dendritic growth which causes stability problems with lithium batteries.
The trick used with the new cathode is to expand titanium disulfide to then introduce magnesium chloride rather than breaking magnesium chloride bonds and storing metallic magnesium alone. The retention of the MgCl bond doubles the charge of the cathode. The researchers created an open nanostructure by extending the gaps in the titanium disulfide by 300% using organic pillars. The resulting gaps were extended to 1.8 nm to insert the magnesium chloride.


Discussion (8 comments)