Solid-state battery breakthrough
on
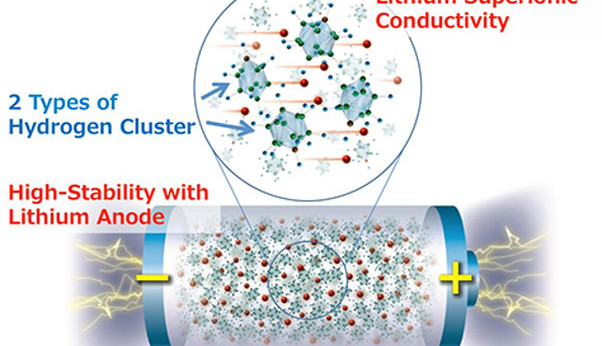
The new material is produced by building structures of hydrogen clusters (complex anions), which shows high stability against metallic lithium, making it an ideal material for the anode of a solid state battery. Such rechargeable batteries with lithium-metal anodes could offer drastically increased energy density levels compared to conventional lithium-ion batteries. Until now the high lithium-ion transfer resistance, caused mainly by the solid electrolyte to metallic lithium instability problem has been the main stumbling block to producing a viable battery design.
The novel electrolyte offers both high ionic conductivity and high stability against metallic lithium which promises to be a real breakthrough in the development of solid-state batteries using a metallic lithium anode.
We need a better battery
Current lithium-ion batteries suffer from limited energy density and can leak electrolyte when misused, creating a fire hazard. An entirely solid-state battery, however promises to offer a better solution for energy storage in the future. Of all known anode materials metallic lithium is considered to be the ultimate material for use in solid-state batteries as it has the highest theoretical capacity (3860 mAh g-1) and the lowest potential -3.04 V (compared with the standard hydrogen electrode).A significant ingredient in a solid-state battery design is the Lithium-ion-conducting solid electrolyte. Its properties determine the ionic conductivity and stability of the solid electrolyte and significantly influence battery performance. Up until recently the main problem with most solid electrolytes has been their chemical/electrochemical instability and/or poor physical contact with metallic lithium which generates side reactions at the interface. These so-called chemical side reactions create by-products, increasing the battery’s interfacial resistance which further degrades with increasing charge cycles.
Previous attempts to get round the problem by alloying lithium with other metals and modifying the interface were not successful. The problem is due to the high thermodynamic reactivity of the lithium metal anode with the electrolyte. The biggest challenges to using metallic lithium anodes are high stability and high lithium-ion conductivity of the solid electrolyte.
The use of complex hydrides with the lithium metal anode has never been tried as a practical battery configuration because of their low ionic conductivity. Using complex hydrides, however achieves high conductivity of lithium ions at room temperature. The research results are published in the journal Nature Communications under the title ‘A Complex Hydride Lithium Super Ionic Conductor for High-Energy-Density All-Solid-State Lithium Metal Batteries’.


Discussion (0 comments)